
Pharma & Life Sciences
AI for Pharma and Life Sciences
Unify knowledge, automate expert reasoning, and operationalize AI across Medical, Commercial, Regulatory, Safety, R&D, and Technical Operations — with full GxP compliance and provenance.
FDA Clinical Decision Support Alignment
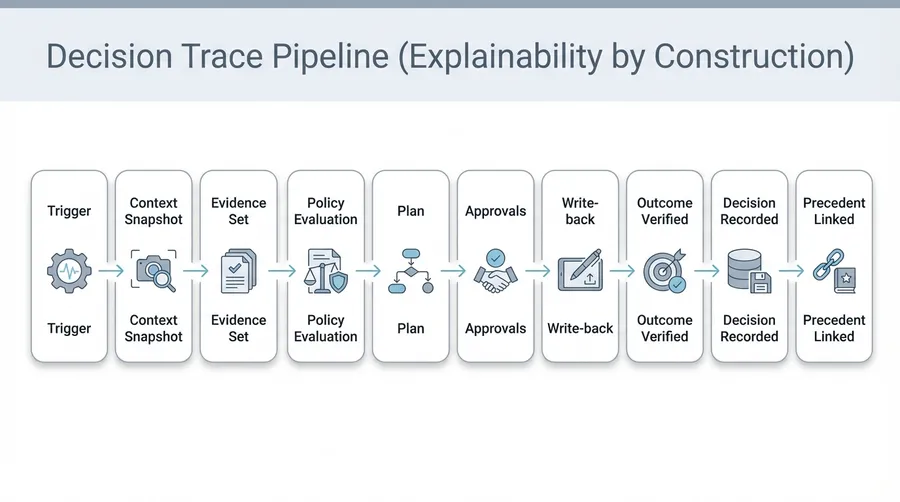
Empower AI includes a productized Clinical CDS Mode designed for alignment with the FDA's Clinical Decision Support software guidance. CDS Mode exemplifies how governed agentic orchestration — decision traces, basis packages, policy gates, and safe write-backs — applies specifically to clinical pharma.
CDS Lane Classifier
Automatically classifies each workflow as Non-Device or Device-lane based on input types and use case characteristics.
Basis Package Generation
Every recommendation is accompanied by a structured "glass-box" Basis Package containing patient-specific inputs, citations to evidence sources, plain-language logic summary, and model provenance.
Decision Trace Capture
The system records the full pragmatic trace: user question, clarification questions, resolved context, accept/modify/reject actions, approvals, and exception routing.
Where Empower AI Transforms Pharma
Beyond clinical decision support, Empower AI delivers high-value impact across regulated, knowledge-heavy pharmaceutical functions.

Medical Affairs
- Automated evidence synthesis from literature and clinical studies
- KOL intelligence — publications, networks, and trial participation
- MSL pre-call dossiers and compliant scientific narratives
- MLR-ready content with citations and full provenance
Commercial & Field
- Pre-call planning with formulary, safety, and competitor intelligence
- Post-call summaries with insights and suggested actions
- Segmentation and territory opportunity detection
- AI-driven field enablement across the sales cycle
Market Access & HEOR
- Payer policy monitoring and coverage intelligence
- AMCP dossier drafting and value narrative assembly
- Real-time HEOR synthesis across trials and economic models
Regulatory Affairs
- Regulatory intelligence across FDA, EMA, and PMDA guidance
- Submission module drafting with traceable evidence chains
- Label change impact analysis and document redlining
- End-to-end provenance for compliance
Pharmacovigilance & Safety
- Adverse event extraction from emails, PDFs, call transcripts
- Signal detection via multi-graph correlation
- Automated case narratives and SOC/HLT mapping
- Continuous monitoring with explainable reasoning trails
Quality & Manufacturing
- SOP ingestion with step-wise troubleshooting guidance
- CAPA draft generation with reasoning trails
- Equipment diagnostics and failure-mode reasoning
- Unified access to QMS, LIMS, batch records
Why EnPraxis AI for Pharma
| Capability | What It Means for Pharma |
|---|---|
| Domain-validated ontologies | Trust and explainability built on pharma-specific knowledge structures |
| Multi-modal ingestion | PDFs, PPTs, Excel, transcripts, SOPs, scientific literature — far exceeds typical RAG |
| Provenance-backed reasoning | Suitable for GxP and regulated workflows with full audit trails |
| Agentic workflows | Orchestrate tools, systems, and reasoning steps across business units |
| AI-native platform | Not bound by CRM object limitations or vendor ecosystem constraints |
| Rapid vertical app generation | Weeks vs. months for purpose-built, domain-specific assistants |
Ready to see it in action?
See how Empower AI can transform your pharmaceutical operations.